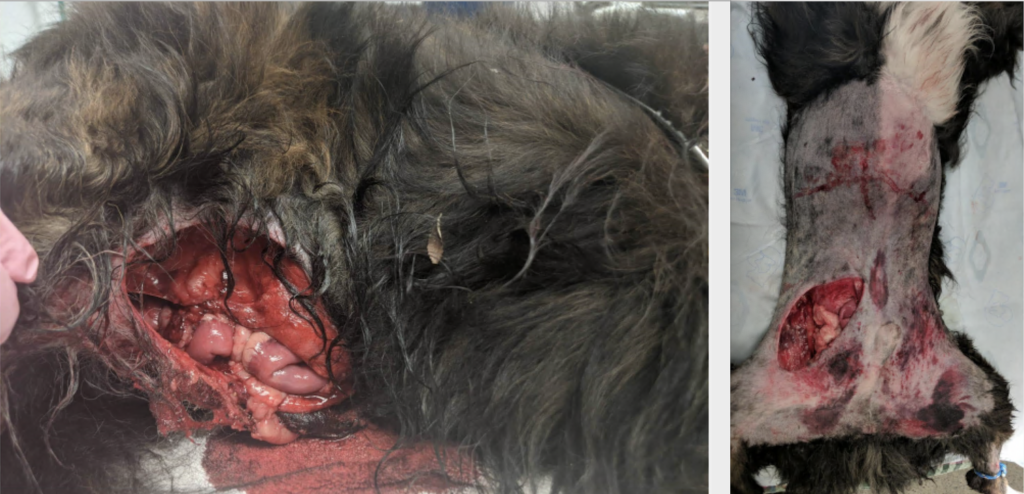
An 8-year-old male castrated Border Collie was presented to the emergency service after a motor vehicle accident. On presentation, he had signs of circulatory shock and a caudal abdominal wall trauma with evisceration of small intestines (Figure 1).
Thoracic point-of-care ultrasound (T-POCUS) and radiographs were suggestive of pulmonary contusions, and bloodwork showed PCV/TS of 63%/6.4 g/dl, hyperlactatemia of 4.4 mmol/l, and mild thrombocytopenia. Preoperative PT/aPTT were within normal limits. The dog was resuscitated with crystalloids and received methadone with broad-spectrum antibiotics prior to surgery. Within 2 hours from hospital admission, the canine patient was taken directly to surgery for abdominal exploration where a complete rupture of his prepubic tendon and protrusion of intraabdominal fat and intestines into a body wall hernia were found. The dog also had marked physiologic degloving of his body wall and inguinal regions that manifested as extensive skin detachment from the thoracic and abdominal walls (the Morel-Lavallée lesion). His abdomen was flushed with sterile saline, the prepubic tendon was repaired, and the body wall was closed. Due to the presence of extensive physiologic degloving, a vacuum-assisted wound closure foam was placed within the skin defect (VAC), and iodine and Hypafix adherent dressing were used to create a negative pressure seal over the VAC foam and skin defect (Figure 2).
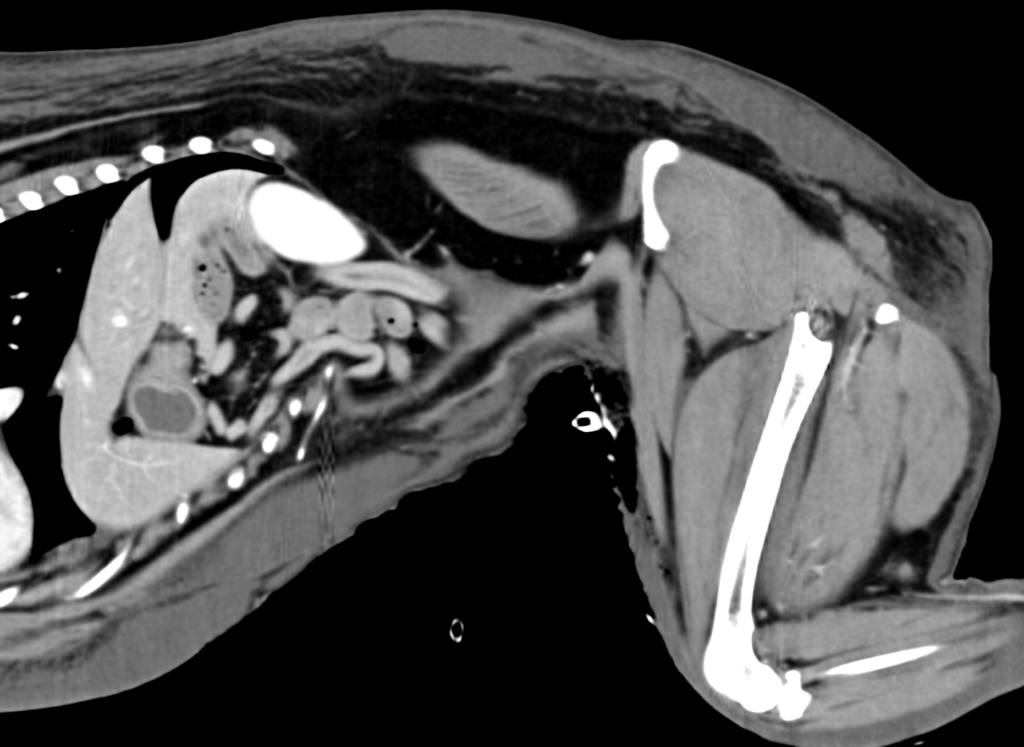
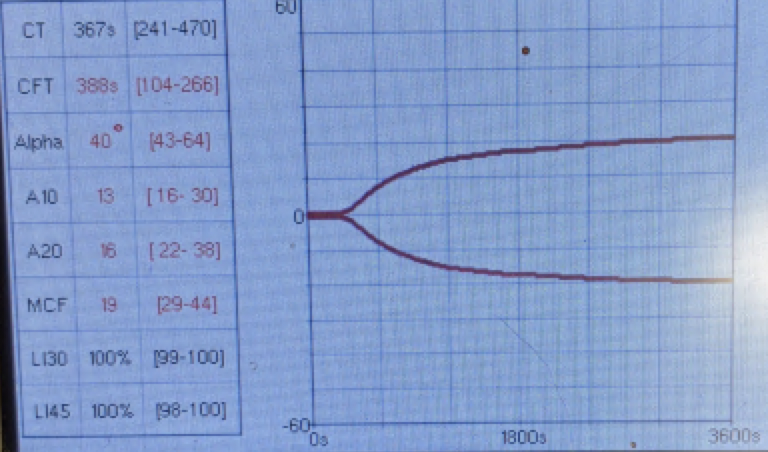
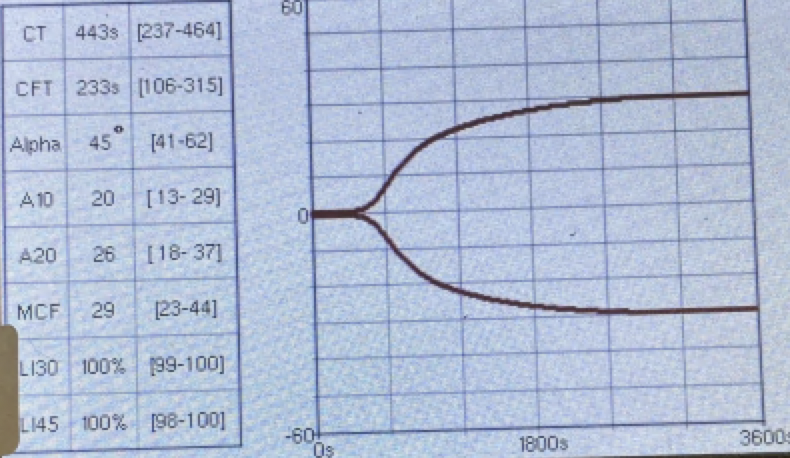
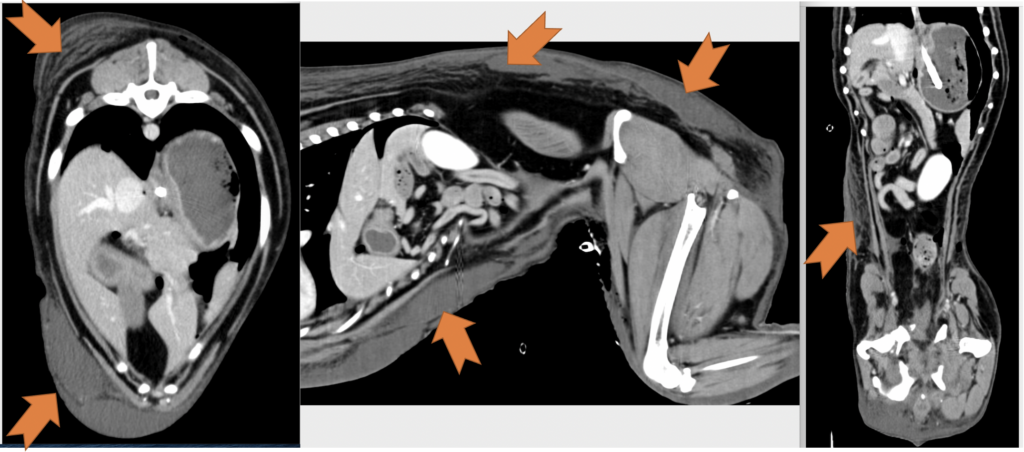
Postoperatively, the patient’s PCV/TS dropped to 24%/3.5 g/dl, and his viscoelastic testing (VCM VetTM) revealed hypocoagulable tracing (Figure 3 and 4). The dog received several units of fresh frozen plasma and whole blood due to ongoing hemodynamic instability. The patient also developed severe ecchymosis and peripheral edema on his ventral thorax, abdomen, perineum, and pelvic limbs. Due to the non-ambulatory state and possibility of spinal trauma, thoracic and abdominal CT were performed 48 hours after presentation. The CT revealed widespread fluid accumulation in the subcutaneous tissues consistent with the Morel-Lavallée lesion and sacrocaudal luxation that did not require surgical management (Figure 5).
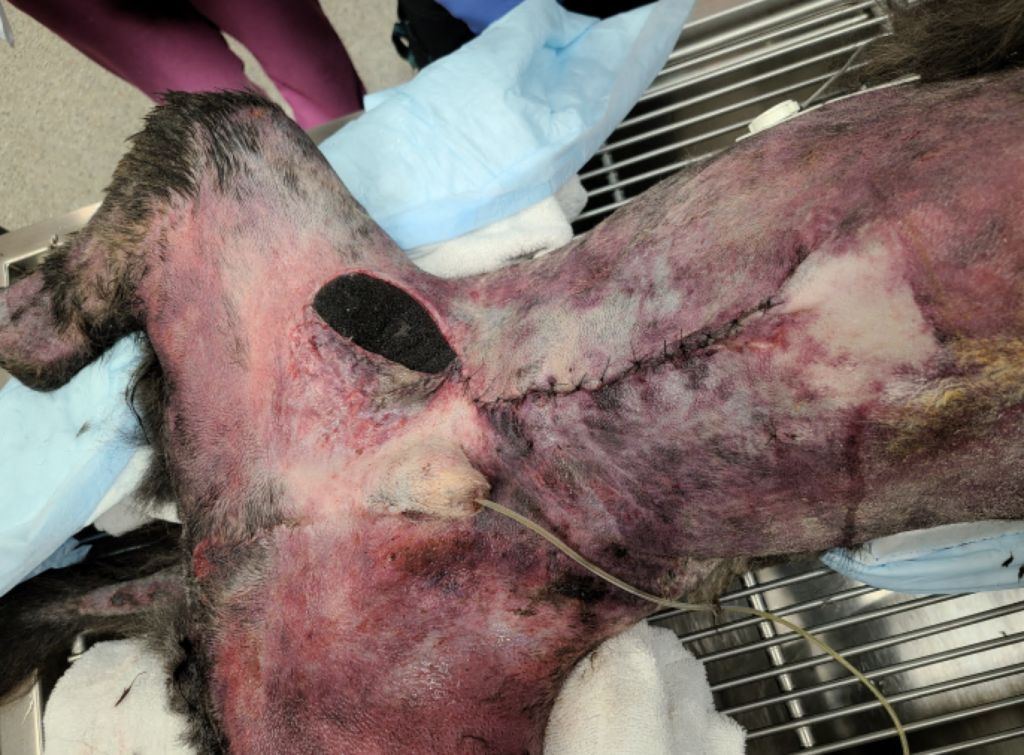
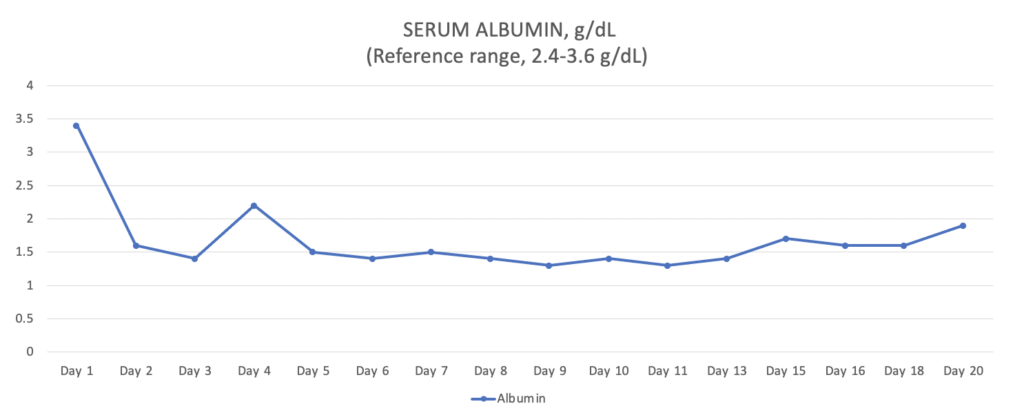
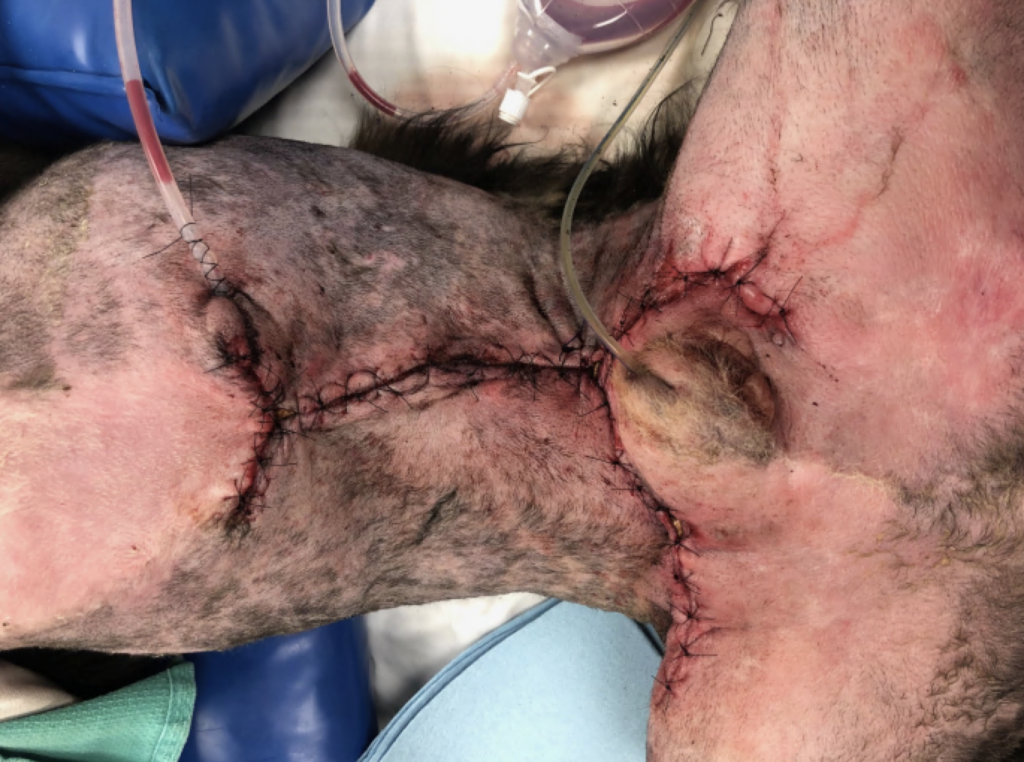
The total duration of ICU hospitalization was 20 days. Over the first 2 weeks, the ventral thoracic and abdominal wall physiologic degloving injury resulted in massive ongoing protein (Figure 6) and fluid losses through the VAC system leading to significant prerenal oliguria (as low as 0.3 ml/kg/hr). Due to the fact that crystalloid fluids led to only partial improvement of oliguria, canine albumin was used. The dog required intensive wound care, vasopressor therapy, nasogastric nutrition, and multiple transfusions (FFP, pRBCs, canine albumin). Around 14 days after the initial injury, he started to show significant improvement, and his wound was completely closed (Figure 7). Two days later, his abdominal drain and urinary catheter were removed. Additionally, he was weaned off of all IV medications and was comfortable on only oral medications. On the 20th day of his hospitalization, he was deemed ready for discharge for monitoring and continued supportive care at home. He regained his ability to walk and completely recovered from all his injuries.
Case Discussion
Morel-Lavallée lesions (MLLs) can occur in polytrauma patients alongside multiple and complex skeletal injuries (Singh et al. 2018). MLLs are characterized as hemolymphatic fluid accumulation between the fascial planes that develops post-soft tissue injury (Bonilla-Yoon et al. 2014; Scolaro et al. 2016; Diviti et al. 2017), and they are described as a closed degloving injury commonly associated with high-energy trauma and shearing forces (McGhie et al. 2018; Scolaro et al. 2016; Diviti et al. 2017). Separation of the superficial and deep fascia results in injury to rich vascular and lymphatic supply leading to accumulation of blood and lymph in this space. With time, a capsulated lesion lined by fibrous capsule may develop, which is filled with blood products, necrotic fatty tissue, debris and fibrin (Bonilla-Yoon et al. 2014; Diviti et al. 2017). MLLs may present as painful fluctuant swelling in the involved sites, and may be missed at initial evaluation and present weeks to month after initial trauma (Diviti et al. 2017).
In the veterinary literature, references to MLLs are limited to two feline case reports (Togni et al. 2015; McGhie et al. 2018), however, to the best of author’s knowledge, there have not been published any canine case reports. McGhie et al. paper discussed the possible difference between the human and feline/canine anatomy that may make people more susceptible to these lesions: “The subdermal plexus is the major vascular supply to the skin, but the orientation of the vessels is notably different between people and dogs and cats (Pavletic 2010). Humans have perforator musculocutaneous and direct cutaneous vessels running perpendicular to the skin. This may make them more prone to damage by shearing forces, in comparison to cats and dogs, where the direct cutaneous arteries of the hypodermis travel parallel to the skin surface”(Pavletic 2010).
In McGhie et al paper, the authors described a 3-year-old male neutered domestic shorthair cat that sustained a severe sacrococcygeal luxation postmotor vehicle trauma. Six days later, a fluid-filled swelling was noticed over the caudoventral abdomen and inguinal regions. The healthcare team attempted a needle aspiration, closed suction drain placement and debridement with subcutaneous tacking, however this treatment failed to resolve the continued accumulation of serosanguinous lymphocyte-rich fluid in the subcutaneous pocket. The cat achieved clinical resolution after surgical debridement and omentalization of the lesion.
Diagnosis of MLLs is made based on a history of trauma, physical examination, lesion distribution and diagnostic imaging. The diagnosis in the described-above canine patient was not challenging, because the MLL lesion was apparent during the surgical exploration, however surgery is not always performed in patients with MLLs. Various imaging modalities including ultrasonography, CT and MRI can be used to diagnose and characterize Morel-Lavallee lesions (Bonilla-Yoon et al. 2014; Diviti et al. 2017). CT imaging findings include internal complexity and a fluid/fluid layer reflecting of internal settling of serosanguinous elements admixed with lymphatic fluid. CT density is usually lower than simple hematomas due to mixing of low density lymphatic fluid and averages from 15-40 Hounsfield units (Diviti et al. 2017; McKenzie et al. 2016).
Many strategies for the treatment of Morel-Lavallee lesions have been reported in human medical literature and there are no established treatment modalities for patients with MLLs. First line therapies, in patients with acute, small lesions without underlying fractures usually include compression bandage application, pain medications, physiotherapy and absolute bed rest (Tejwani et al. 2007; Diviti et al. 2017). Percutaneous drainage can be used for larger acute lesions. It may also be attempted along with sclerotherapy in patients with chronic lesions (Tseng et al. 2006; Diviti et al. 2017). Open debridement as a first line therapy has been attempted in patients with acute lesions with underlying open fractures and chronic lesions with evidence of infection or tissue necrosis due to a local mass effect (Kumar et al. 2015). In patients who are refractory to percutaneous drainage with sclerotherapy, open debridement may also be considered as the final therapy (Diviti et al. 2017).
The Bottom Line
Small animal practitioners should be aware of Morel-Lavallee lesions and treatment challenges they might present. Patients with vehicular polytraumas are at highest risk for MLLs. Significant protein loss and slow recovery requiring prolong hospitalization may lead to very expensive treatment and high rates of euthanasia in this population of animals.
References
- Singh R, Rymer B, Youssef B, Lim J. The Morel-Lavallée lesion and its management: A review of the literature. J Orthop. 2018 Aug 28;15(4):917-921.
- Bonilla-Yoon I, Masih S, Patel DB, et al. The Morel-Lavallee lesion: pathophysiology, clinical presentation, imaging features, and treatment options. Emerg Radiol 2014; 21: 35–43.
- Scolaro JA, Chao T and Zamorano DP. The Morel-Lavallée lesion: diagnosis and management. J Am Acad Orthop Surg 2016; 24: 667–672.
- Diviti S, Gupta N, Hooda K, et al. Morel-Lavallee lesions – review of pathophysiology, clinical findings, imaging findings and management. J Clin Diagn Res 2017; 11: TE01–TE04.
- McGhie JA, Gibson ID, Herndon AM. Morel-Lavallée lesions: a phenomenon in cats? Case report and review of the literature. JFMS Open Rep. 2018 May 22;4(1)
- Togni A, Sievert C, Hurter K, et al. Chronic expanding haematoma in a cat. J Feline Med Surg 2015; 17: 733–736.
- Pavletic M. Atlas of small animal wound management and reconstructive surgery. 3rd ed. St Louis, MO: WB Saunders, 2010.
- McKenzie GA, Niederhauser BD, Collins MS, Howe BM. CT characteristics of Morel-Lavallée lesions: An under-recognized but significant finding in acute trauma imaging. Skeletal Radiol. 2016;45(8):1053-60.
- Tejwani SG, Cohen SB, Bradley JP. Management of Morel-Lavallee lesion of the knee: Twenty-seven cases in the national football league. Am J Sports Med. 2007;35(7):1162–67.
- Tseng S, Tornetta P 3rd. Percutaneous management of Morel-Lavallee lesions. J Bone Jt Surg Am. 2006;88(1):92–96.
- Kumar S, Hasan R, Kadavigere R, Maddukuri SB, Puppala R. Morel-Lavallee Lesion (MLL) Mimicking a soft tissue neoplasm. J Clin Diagn Res. 2015;9(4):TD01- 02.
