Introduction
This guideline created by the VETEMCRIT Academy is based on the most current veterinary and human medical literature, encompassing both adult and pediatric research. It was designed as a practical clinical tool for small animal veterinary practitioners.
What is HHS?
HHS (Hyperosmolar Hyperglycemic State) is a form of diabetic crisis characterized by severe hyperglycemia (>600 mg/dL or >33 mmol/L), minimal or absent urine/plasma ketones, and serum osmolality greater than 325 mOsm/kg in dogs or 350 mOsm/kg in cats (Koenig et al. 2022).
Diabetic Ketoacidosis (DKA) and HHS are both forms of diabetic crises. They can be viewed as different manifestations along the same spectrum, as evidenced by the fact that some veterinary and human patients present with a combination of HHS and DKA.
However, there are distinct features of HHS that differentiate it from DKA. Veterinarians must be aware of these differences, as they may impact clinical management.
Key Differences Between HHS and DKA
1. Insulin Deficiency and Ketogenesis: While DKA is a state of near absolute insulin deficiency, there is enough insulin present in HHS to prevent lipolysis and ketogenesis but not enough to facilitate glucose utilization. Therefore, patients with DKA develop profound ketonemia leading to severe high anion gap metabolic acidosis, whereas animals with HHS do not.
- It takes one-tenth as much insulin to suppress lipolysis as it does to stimulate glucose utilization by muscles and liver. Thus, patients with HHS have just enough insulin to prevent ketogenesis but not to control hyperglycemia (Gosmanov et al. 2021).
2. Renal Dysfunction and Hyperglycemia: The most important pathophysiologic factor that differentiates HHS from DKA, aside from the lack of significant ketogenesis, is the profound osmotic diuresis leading to the reduction in glomerular filtration rate (GFR). Renal dysfunction, whether prerenal or renal, leads to worsening hyperglycemia, sometimes exceeding 1000-1500 mg/dL (55-80 mmol/L) that is driving the osmotic diuresis even further.
- Normally, all glucose that exceeds the renal threshold is excreted in the urine. However, this mechanism is significantly impaired in animals with reduced GFR, allowing severe hyperglycemia (exceeding 600 mg/dL = 33 mmol/L) to develop. This drives osmotic diuresis, further worsening renal function in a vicious cycle:
- Low GFR → worsening hyperglycemia → osmotic diuresis → hypovolemia → low GFR → worsening hyperglycemia.
3. Insulin Requirements and Management: Since patients with HHS do not accumulate excessive ketone bodies (unless it is a case of combined HHS/DKA), their insulin requirements are lower, and normalization of the intravascular volume and GFR is the number one priority in their initial management.
How common is HHS in dogs and cats?
HHS accounted for 6.4% of total emergency room visits by diabetic cats (Koenig et al. 2004), and 5% of ER visits by diabetic dogs (Trotman et al. 2013).
Table 1. Diagnostic criteria of HHS vs. DKA vs. mixed HHS/DKA
| Parameters | HHS | DKA | HHS+DKA |
| Hyperglycemia | >600 mg/dl (>33 mmol/L) | 250-600 mg/dl (~14-33 mmol/L) | >600 mg/dl (>33 mmol/L) |
| Ketonemia via portable ketone meter (BHB* levels) | <2.5-3 mmol/L | >2.5-3 mmol/L | >2.5-3 mmol/L |
| Ketonuria** | Absent to trace | Mod-severe | Mod-severe |
| Calculated osmolality***, mOsm/kg | >325 in dogs, >350 in cats | <325-350 | >325 in dogs, >350 in cats |
| pH/HCO3 | Usually, >7.3/>15, however, may develop HAGMA**** from severe uremia/hyperlactatemia | <7.3/<15 | <7.3/<15 |
| Azotemia | Present in large proportion of cases | +/- | Present in large proportion of cases |
| Dehydration/hypovolemia | Usually, severe | Mild to severe | Usually, severe |
| Corrected plasma sodium level | Usually, elevated | Low to normal | Usually, elevated |
*BHB = beta-hydroxybutyrate, the predominant ketone body in dogs and cats
**Ketonuria = While ketonuria is present in 94% to 100% of dogs at the time of DKA diagnosis, urine dipsticks may not always reliably detect it (Di Tommaso et al., JVIM 2009). This is because dipstick tests primarily measure acetoacetate and, to a lesser extent, acetone, but not BHB.
***Calculated osmolality (USA units, i.e. BUN and glucose measured in mg/dl): (2 x Na+ + BUN [mg/dL] /2.8 + glucose [mg/dl] /18). Alternatively, effective osmolality can be calculated: 2 x Na+ + glucose/18 can be used (without BUN)
Calculated osmolality (International units, i.e. BUN and glucose measured in mmol/L): (2 x Na+ + BUN [mmol/L] + glucose [mmol/L]). Alternatively, effective osmolality can be calculated: 2 x Na+ + glucose can be used (without BUN)
****HAGMA = High Anion Gap Metabolic Acidosis: Usually, caused by accumulation of the following anions: ketones, uremic acids, toxins (e.g. ethylene glycol, methanol), lactic acid.
Aims of therapy
- Improvement in clinical status and replacement of all estimated fluid losses by 24 hours leading to improved GFR and urine output
- Gradual decline in hyperglycemia (target goal: 50-75 mg/dl/hour = 2.5-4 mmol/L/hr, in some references – up to 100 mg/dl/hr = 5.5 mmol/L/hr), hypernatremia (target goal: 10-12 mmol/kg/day ~0.5 mmol/L/hr) and serum osmolality (3-8 mOsm/kg/hr).
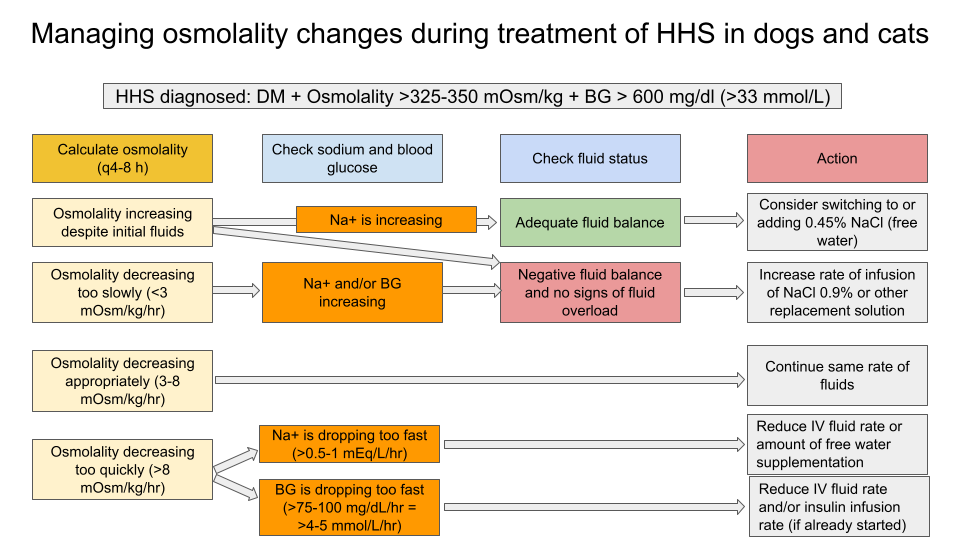
- The overall rate of decrease in osmolality is more important than the individual decreases in sodium or blood glucose levels.
- Avoid hypoglycemia, hypokalemia and hypophosphatemia.
- Prevention of potential complications such as cerebral edema (caused by acute drop in plasma osmolality) or osmotic demyelination syndrome (caused by acute increase in plasma osmolality)
- The key parameter in HHS is osmolality. Sodium and glucose are the main contributors to this, and too rapid changes are dangerous because large fluid shifts can lead to neurological complications, in particular cerebral edema and osmotic demyelination.
HHS resolution criteria
- Serum osmolality <325 mOsm/kg in dogs and <350 mOsm/kg in cats
- Hypovolemia has been corrected
- Urine output >0.5-1 ml/kg/hr (if measured)
- Mental status returned to pre-morbid state
- Blood glucose <300 mg/dl (<15-17 mmol/L)
Table 2. Main differences in management of HHS vs. DKA vs. mixed HHS/DKA
| Parameters | HHS | DKA | HHS+DKA |
| Treatment of hyperglycemia | Decrease in BG should not exceed 50-75 mg/dl/hr to prevent cerebral edema | The rate of hyperglycemia resolution is less important | Same as HHS |
| Plasma osmolality monitoring | Decrease in plasma osmolality should not exceed 8 mOsm/kg/hr | Plasma osmolality should remain normal | Same as HHS |
| Insulin therapy | Insulin therapy should be delayed. High glucose level helps the maintenance of intravascular volume therefore aggressive insulin treatment and a rapid drop in plasma glucose can lead to circulatory collapse; insulin supplementation is given at lower rates than in DKA because there is minimal ketosis; recommended insulin infusion rate is 0.025-0.05 U/kg/hr titrated to ensure the drop in BG 50-75 mg/dl/hr = 2.5-4 mmol/L/hr (in some cases, rate may need to be increased to 0.1 U/kg/hr) | Insulin therapy is crucial for resolution of ketoacidosis; should not be delayed for longer than 6-8 hours; infusion rate may be equal to or exceed 0.1 U/kg/hr | Same as DKA |
| Fluid type | Due to the lack of acidemia and high prevalence of hypernatremia, NaCl 0.9% can be used initially. Free water supplementation will likely be required to treat hypernatremia (0.45% NaCl is most commonly recommended as a free water source in human pediatric and adult guidelines) | Balanced replacement solutions (e.g. LRS, Norm-R) should be prioritized because NaCl 0.9% will further promote acidosis due to the high chloride content) | Depends on electrolyte derangements and degree of acidemia |
| Fluid rate and urine output monitoring | Owing to severe osmotic diuresis, urine output monitoring is more important than in DKA to guide total fluid rate | May depend on other comorbidities and renal function | Same as HHS |
Management timeline and priorities
First 60 min from admission: Determine clinical status and establish the diagnosis of HHS
- Collect history, perform full physical examination with emphasis on perfusion parameters, mentation, and respiratory status.
- Perform point-of-care ultrasound to rule out cavitary effusions, CHF/cardiogenic shock (LA/Ao ratio, LV contractility, RV/CVC enlargement) and improve diagnosis of shock, respiratory distress, and azotemia if present.
- Establish adequate intravascular lines.
- Perform point-of-care bloodwork – venous blood gas, creatinine, BUN, glucose, electrolytes (Na, K, Cl, Mg and phosphate).
- Start fluid resuscitation to correct hypovolemia (start with 10-20 ml/kg of crystalloid replacement solution in dogs or 5-10 ml/kg in cats))
- Severe osmotic diuresis typically leads to losses of free water exceeding sodium losses resulting in hypertonic dehydration.
- The majority of human adult and pediatric guidelines recommend initial fluid resuscitation (i.e. fluid boluses) with NaCl 0.9% since most HHS patients are hypernatremic, and the use of crystalloids with higher sodium content such as NaCl 0.9% will prevent an acute drop of plasma sodium level.
- Once vital signs have stabilized, replacement fluids of 0.45-0.9% NaCl over 24-48 hours. Veterinary textbooks (expert opinions only) recommend the use of balanced crystalloid solutions (e.g. Normosol-R). No human or veterinary studies comparing different crystalloid solutions in HHS have been performed.
- Calculate baseline serum osmolality:
- Calculated osmolality (USA units, i.e. BUN and glucose measured in mg/dl): (2 x Na+ + BUN [mg/dL] /2.8 + glucose [mg/dl] /18). Alternatively, effective osmolality can be calculated: 2 x Na+ + glucose/18 can be used (without BUN);
- Calculated osmolality (International units, i.e. BUN and glucose measured in mmol/L): (2 x Na+ + BUN [mmol/L] + glucose [mmol/L]). Alternatively, effective osmolality can be calculated: 2 x Na+ + glucose can be used (without BUN)
- Calculate corrected sodium (i.e. expected increase in Na+ after hyperglycemia is corrected) = Na (measured) + 1.6 x [(Measured glucose [mg/dl] – 100) / 100]
- Same calculation but for those of you who measure blood glucose in mmol/L:
Na (measured) + 1.6 x [(Measured glucose [mmol/l] – 5.5) / 5.5)
- For example, if patient’s Na+=135 and blood glucose = 600 mg/dl (=33.3 mmol/L), then you expect that corrected Na+ will become 143, i.e. it will go up by ~8 mmol/l (from 135 to 143 mg/dl) AFTER you decrease patient’s BG from 600 to 100 mg/dl (from 33 mmol/L to 5.5 mmol/L).
- Order full CBC, chemistry and urinalysis.
1-6 hours
- Continue cardiovascular/mentation/respiratory monitoring q1-2 hours.
- Discuss admission to the ICU with the critical care team.
- Strongly consider urine output monitoring and sampling line placement (central venous catheter or peripherally-inserted central catheter – a PICC line).
- Continue fluid therapy to correct fluid deficit and account for ongoing losses (osmotic diuresis +/- vomiting/diarrhea) and maintenance requirements.
- If hypernatremia is present, calculate a free water deficit and start supplementation of free water (e.g. 0.45% NaCl OR dextrose 5% if BG is dropping too fast OR regular water via NG tube if the patient tolerates it without vomiting and other options are not ideal – for example, in animals with CHF – see hypernatremia treatment guidelines for more information).
- Withhold insulin therapy as long as BG drops by 50-75 mg/dl/hour with fluid therapy only.
- Fluid replacement alone with 0.9% sodium chloride solution will result in a falling blood glucose. Lowering glucose level by early insulin use in HHS will lower osmolality precipitously and risks osmotic fluid shifts leading to circulatory collapse. There is also higher risk of hypokalemia and hypoglycemia.
- Further assess for precipitating causes: sepsis, acute pancreatitis, AKI/CKD/pyelonephritis, CHF, UTI, Cushing’s disease, etc.
- Monitor BG every 1-2 hours (via sampling line, Freestyle Libre, or ear pinna/paw pricks if patient tolerates it and other options are not available).
- Monitor K+ and Na+ every 4-6 hours, Mg and PO4 every 8-12 hours.
- Monitor serum osmolality every 4-6 hours.
- Monitor body weight every 4-8 hours to aid in fluid balance management (e.g. if a dehydrated patient is not gaining weight, this means that the ongoing losses are exceeding fluid intake).
- Start correcting hypokalemia, hypophosphatemia and hypernatremia if present
- During insulin treatment and rehydration, serum potassium +/- phosphorous levels fall rapidly; therefore, it is recommended that potassium replacement should be initiated before insulin is started (unless the patient is hyperkalemic at baseline)
- A free online potassium supplementation calculator can be found here.
- The correction of severe hyperglycemia will lead to increasing sodium levels due to the fluid shift from extravascular to intravascular compartment, however an overall change in plasma osmolality is more important (see Figure 1 for plasma osmolality management guidelines).
- Monitor blood ketones, creatinine/BUN every 12-24 hours.
6-12 hours
- Continue cardiovascular/mentation/respiratory/urine output monitoring q1-2 hours.
- Reassess hydration level / urine output and adjust fluid therapy to correct fluid deficit and account for ongoing losses (osmotic diuresis +/- vomiting/diarrhea) and maintenance requirements.
- Once the glucose has ceased to fall following initial fluid resuscitation, reassessment of fluid intake and evaluation of renal function must be undertaken. Insulin may be started at this point.
- Consider starting insulin therapy once the following criteria are met:
- BG >250 mg/dl and is dropping by less than 50 mg/dl/hr
- K+ >3.5 mmol/L
- Phosphate >2.5 mg/dl
- Insulin can be administered via an intermittent IM or IV CRI protocols:
- IM: administer 0.1 U/kg of regular insulin, then 0.05 U/kg q2-4h to reduce BG by 50-75 mg/dl/hr and maintain BG within 100-300 mg/dl range. The insulin dose may be increased by 20-25% at a time if BG fails to decrease.
- IV: Consider either fixed-rate (preferred in human guidelines at the 0.025-0.05 U/kg/hr rate) or variable-rate infusion protocol (see below). Note: Animals with HHS (without co-existing DKA) may require lower rate of insulin and constant insulin supplementation is less critical, therefore their hourly rate may be reduced from 0.09-0.1 U/kg/hr (DKA CRI protocols) to 0.025-0.05 U/kg/hr.
Option #1: Variable-rate insulin infusion (VRI) protocol adopted for veterinary HHS patients (i.e. lower insulin infusion rate as compared with the original DKA protocol):
Three bags are pre-made:
Bag #1: 1.1 U/kg of insulin added to a 250 ml bag of NaCl 0.9% for both dogs and cats.
Bag #2: An isotonic crystalloid solution, used to correct dehydration and provide maintenance rates.
Bag #3: A 500 ml isotonic crystalloid solution with 50 ml of dextrose 50% added to create a final 5% dextrose solution.
Fluid and Insulin infusion rates based on blood glucose (BG) concentration:
- BG >15 mmol/L (>270 mg/dL): 10 ml/hr from bag #1; the bag #2 is used to correct dehydration as needed.
- BG 12-14.9 mmol/L (216-269 mg/dL): 7 ml/hr from bag #1 + bag #2 as needed to correct dehydration + bag #3 at 2 ml/kg/hr
- BG 8-11.9 mmol/L (144-215 mg/dL): 5 ml/hr from bag #1 + bag #2 as needed to correct dehydration + bag #3 at 2 ml/kg/hr
- BG 5-7.9 mmol/L (90-143 mg/dL): 5 ml/hr from bag #1 + 4 ml/kg/hr from bag #3 + bag #2 as needed to correct dehydration.
- BG <5 mmol/L (<90 mg/dL): Stop insulin, administer 4 ml/kg/hr from bag #3 + bag #2 as needed
An example of the VRI-HHS protocol table that can be placed as an order on the treatment sheet for a 30-kg dog with HHS (7% dehydrated; serum K+=3.4 mmol/L; phosphate = 0.9 mg/dl).
| Blood Glucose: (mmol/L) [mg/dL] | BAG #1 Insulin Bag (250 ml NaCl 0.9% + 1.1U/kg of regular insulin) | Bag #2 Hydration Bag (995 ml of NaCl 0.9% + KPhos 5.3 ml = 15.8 mM of phosphate = 0.1 mM/kg/hr if run at 190 ml/hr) | Bag #3 Dextrose Bag (895 ml of NaCL 0.9% + 100 ml of Dextrose 50% + KPhos 5.3 ml) | Total fluid rate (bag 1 + bag 2 + bag 3) Rehydration over 12h + Maintenance |
| >15 [270] | 10 ml/hr = 0.045 U/kg/hr | 190 | 0 | 200 |
| 12-14.9 [216 – 269] | 7 ml/hr = 0.03 U/kg/hr | 130 | 60 | 197 |
| 8-11.9 [144-215] | 5 ml/hr = 0.02 U/kg/hr | 65 | 60 | 200 |
| 5-7.9 mmol/L [90-143 mg/dL] | 5 ml/hr = 0.02 U/kg/hr | 65 | 120 | 200 |
| <5 mmol/L [<90 mg/dL] | 0 | 80 | 120 | 200 |
Option #2: Fixed-rate insulin infusion (FRI) protocol adopted for veterinary HHS patients (i.e. lower insulin infusion rates as compared with the original DKA protocol):
Three bags are pre-made:
Bag #1: 25 units of regular insulin added to a 500 ml bag of NaCl 0.9% (0.05 U of insulin per ml of solution)
Bag #2: An isotonic crystalloid solution, used to correct dehydration and provide maintenance rates.
Bag #3: A 500 ml isotonic crystalloid solution with 50 ml of dextrose 50% added to create a final 5% dextrose solution
Fluid and Insulin infusion rates based on blood glucose (BG) concentration:
- BG >15 mmol/L (>270 mg/dL): Insulin from bag #1 at 1 ml/kg/hr (0.05 U/kg/hr) + fluid from bag #2 to correct dehydration and provide maintenance.
- BG 7-15 mmol/L (126-270 mg/dL): Insulin from bag #1 at 1 ml/kg/hr (0.05 U/kg/hr) + fluid from bag #2 (hydration bag) and #3 (5% dextrose) at ≥2 ml/kg/hr
- BG <7 mmol/L (<126 mg/dL): Stop insulin, continue fluid from bag #3 (5% dextrose)
An example of the FRI-HHS protocol table that can be placed as an order on the treatment sheet for a 30-kg dog with HHS (7% dehydrated; serum K+=3.4 mmol/L; phosphate = 0.9 mg/dl).
| Blood Glucose: (mmol/L) [mg/dL] | BAG #1 Insulin Bag (500 ml NaCl 0.9% + 50U insulin) | Bag #2 Hydration Bag (995 ml of NaCl 0.9% + KPhos 5.3 ml = 15.8 mM of phosphate = 0.1 mM/kg/hr if run at 190 ml/hr) | Bag #3 Dextrose Bag (895 ml of NaCL 0.9% + 100 ml of Dextrose 50% + KPhos 5.3 ml) | Total fluid rate (bag 1 + bag 2 + bag 3) Rehydration over 12h + Maintenance |
| >15 [270] | 30 ml/hr = 0.05 U/kg/hr | 190 | 0 | 220 |
| 7-14.9 [126 – 269] | 30 ml/hr = 0.05 U/kg/hr | 130 | 60 | 220 |
| <7 [126] | 0 | 0 | 220 | 220 |
12-24 hours
- Continue cardiovascular/mentation/respiratory/urine output monitoring.
- Reassess fluid balance and hydration, adjust a total fluid rate as needed.
- Monitor VBG every 8-12 hours, Na/K every 4-8 hours, phosphate/Mg every 12-24 hours.
- Monitor BG every 1-2 hours to ensure it is dropping by 50-75 mg/dl/hour, adjust insulin dose/frequency as needed.
- Continue addressing the underlying disease.
24-72 hours
- De-escalate cardiovascular/mentation/respiratory/urine output monitoring if the patient is more stable.
- If the patient is anorexic, consider placing an NG/NE tube or esophagostomy depending on the underlying disease and expected duration of anorexia, the risk of general anesthesia, etc.
- If BG is within 100-300 mg/dl (=5-16 mmol/L), and the patient is still receiving insulin infusion, consider switching to intermittent IM or SQ insulin and de-escalate BG monitoring to q4h.
- If the patient is eating, consider switching to intermediate- or long-lasting insulin injections.
- Reassess fluid balance and hydration, adjust a total fluid rate as needed; discontinue fluids if patient is hydrated and eating.
- De-escalate VBG and electrolytes monitoring if patient is stable and electrolyte derangements have been corrected.
References
- Mustafa OG, Haq M, Dashora U, Castro E, Dhatariya KK; Joint British Diabetes Societies (JBDS) for Inpatient Care Group. Management of Hyperosmolar Hyperglycaemic State (HHS) in Adults: An updated guideline from the Joint British Diabetes Societies (JBDS) for Inpatient Care Group. Diabet Med. 2023 Mar;40(3):e15005. doi: 10.1111/dme.15005. Epub 2022 Dec 21. PMID: 36370077; PMCID: PMC10107355.
- Gosmanov AR, Gosmanova EO, Kitabchi AE. Hyperglycemic Crises: Diabetic Ketoacidosis and Hyperglycemic Hyperosmolar State. 2021 May 9. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. PMID: 25905280.
- Koenig, Amie. “Hyperglycemic Hyperosmolar Syndrome.” In Small Animal Critical Care Medicine, edited by Deborah Silverstein and Kate Hopper, 438-439. Elsevier – Health Sciences Division, 2022. ProQuest Ebook Central, http://ebookcentral.proquest.com/lib/tamum-ebooks/detail.action?docID=7242470.
- Ng, S.M., Edge, J.A., & Timmis, A.E. (2021). Practical Management of Hyperglycaemic Hyperosmolar State (HHS) in children (Version 3, April 2021; Review 2024). Association of Children’s Diabetes Clinicians.
- Koenig A, Drobatz K, Beale AB, King L: Hyperglycemic, hyperosmolar syndrome in feline diabetics: 17 cases (1995-2001), J Vet Emerg Crit Care 14:30, 2004.
- Trotman TK, Drobatz KJ, Hess RS: Retrospective evaluation of hyperosmolar
hyperglycemia in 66 dogs (1993-2008), J Vet Emerg Crit Care 23(5):
557-564, 2013. - Glaser, N., Fritsch, M., Priyambada, L., Rewers, A., Cherubini, V., Estrada, S., Wolfsdorf, J. I., & Codner, E. (2022). ISPAD Clinical Practice Consensus Guidelines 2022: Diabetic ketoacidosis and hyperglycemic hyperosmolar state. Pediatric Diabetes, 23(7), 835-856. https://doi.org/10.1111/pedi.13406

One thought on “Management of Hyperosmolar Hyperglycemic State (HHS) in Dogs and Cats: A Clinical Guideline for Small Animal Practitioners”